2025
42) Ash, J.; Curtis, K.; Bhowmik, S.; Odoh, S. O.; Kang, J. Y. “Hydropersulfide (RSSH)-Mediated Hydrothiolation of Alkenes” J. Org. Chem., 2025, 90, 1167-1174 https://doi.org/10.1021/acs.joc.4c02309

2024
41) Ash, J.; Kang, J. Y. “Synthesis of unsymmetrical phosphorus disulfides” Org. Biomol. Chem., 2024, 24, 7492-7499. https://doi.org/10.1039/D4OB01099J

40) Ash, J.; Ahmed, M.; Le, N.; Huang, H.; Kang, J. Y. “Catalytic, regioselective Friedel–Crafts alkylation of beta-naphthol” New J. Chem., 2024,48, 4224-4228. https://doi.org/10.1039/D3NJ05580A

2023
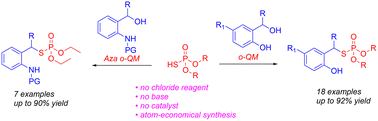
(39) Ash, J.; Kang, J. Y. “Catalyst-free thiophosphorylation of in situ formed ortho-quinone methides” Org. Biomol. Chem., 2023, 21, 2370-2374. DOI:https://doi.org/10.1039/D2OB02169B
2022
(38) Patent Issued
Kang, J. Y.; Huang, H. “Chiral n-heterocyclic phosphorodiamidic acids (NHPAS) and derivatives as novel bronsted acid catalysts” 11,248,014, 2/15/2022
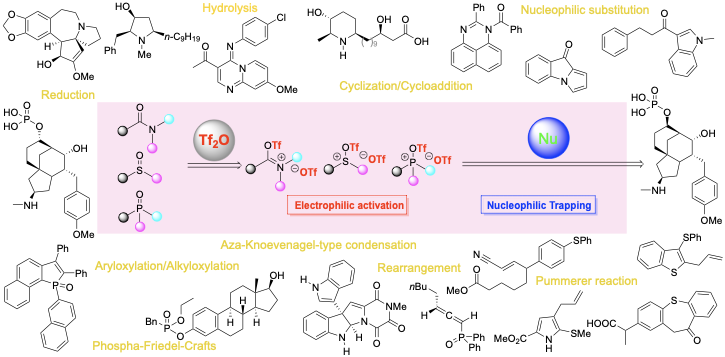
(37) Huang, H.; Kang, J. Y.”Triflic Anhydride (Tf2O)-Activated Transformation of Amides, Sulfoxides and Phosphorus Oxides via Nucleophilic Trapping” Synthesis, 2022, 54, 1157-1202. DOI: 10.1055/a-1679-8205 (Invited SYNTHESIS REVIEW article)
2021
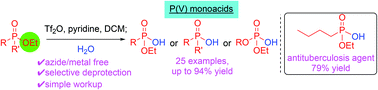
(36) Ash, J.; Huang, H.; Cordero, P.; J. Y. Kang “Selective hydrolysis of phosphorus(v) compounds to form organophosphorus monoacids” Org. Biomol. Chem. 2021, 19, 6007-6014 doi.org/10.1039/D1OB00881A
(35) Patent Issued
Kang, J. Y.; Huang, H. “Metal-free direct arylation of dialkyl phosphonates for the synthesis of mixed alkyl aryl phosphonates” 10,927,135, 2/23/2021
2020
(34) Patent Issued
Kang, J. Y.; Mulla, K.; Aleshire, K.; Forster, M P. “N-heterocyclic phosphines” 10,633,403, 4/28/2020
(33) Le Pham, N. S.; Shin, H.; Kang, J. Y.; Sohn, J. H. “Oxidative Dehydrosulfurative Cross-Coupling of 3, 4-Dihydropyrimidine-2-thiones with Alkynes for Access to 2-Alkynylpyrimidines” J. Org. Chem. 2020, 85, 5087-5096. https://doi.org/10.1021/acs.joc.0c00091
2019
(32) Patent Issued
Kang, J. Y.; Huang, H.; Molleti, N. “Functionalized phosphonates via Michael addition”,10,435,423, 10/8/2019
(31) Ash, J.; Huang, H.; Kang, J. Y. “Metal- and Chloride Reagent-free Synthesis of Mixed Thiophosphates” Org. Biomol. Chem.2019, 17, 3812-3818. DOI: 10.1039/c9ob00370c
(30) Huang, H.; Kang, J. Y.”New Strategies for Activation of Phosphonates/Phosphates to Forge Functional Phosphorus Compounds” Synlett, 2019, 30, 635-641. DOI: 10.1055/s-0037-1611699 (Invited SYNPACTS article)
Jun Yong Kang: Thieme Chemistry Journal Awardee in 2019
2018
(29) Patent Issued
Kang, J. Y.; Mulla, K. “Methods and Compositions for Substituted Alpha-Aminophosphonate Analogues”, US10087204B2, 10/2/2018
(28) Huang, H.; Ash, J.; Kang, J. Y. “Tf2O‐Promoted Activating Strategy of Phosphate Analogues: Synthesis of Mixed Phosphates and Phosphinate” Org. Lett. 2018, 4938-4941, DOI: 10.1021/acs.orglett.8b02073
- Highlighted on the Organic Chemistry Portal. [link]
(27) Huang, H.; Ash, J.; Kang, J. Y. “Base-Controlled Fe(Pc)-Catalyzed Aerobic Oxidation of Thiols for the Synthesis of S-S and S-P(O) Bond” Org. Biomol. Chem. 2018, 16, 4236-4242, http://dx.doi.org/10.1039/C8OB00908B
(26) Le, N.; Huang, H.; Kang, J. Y. “One-pot synthesis of phosphorodiamidothioates using N-heterocyclic phosphine (NHP)-thiourea” Tetrahedron Lett. 2018, 56, 2296-2298, https://doi.org/10.1016/j.tetlet.2018.04.080
(25) Huang, H.; Zhu, H.; Kang, J. Y. “Regio- and Stereoselective Hydrophosphorylation of Ynamides for the Synthesis of β-Aminovinylphosphine Oxides” Org. Lett. 2018, 20, 2778-2781, DOI: 10.1021/acs.orglett.8b01065
(24) Huang, H.;, J.; , , H.; Kang, J. Y. “Direct Aryloxylation/Alkyloxylation of Dialkyl Phosphonates for the Synthesis of Mixed Phosphonates” Angew. Chem. Int. Ed.2018, 57, 6624-6628, http://dx.doi.org/10.1002/anie.201802082
(23) Shetty, M.; Huang, H.; Kang, J. Y. “Regioselective Synthesis of α- and γ-Amino Quinolinyl Phosphonamides Using N-Heterocyclic Phosphines (NHPs)“Org. Lett. 2018, 20, 700-703, DOI: 10.1021/acs.orglett.7b03829.
2017
(22) Huang, H.; Kang, J. Y. “Organocatalytic Phosphonylation of In Situ Formed o-Quinone Methides”Org. Lett.2017, 19, 5988-5991, DOI: 10.1021/acs.orglett.7b03019.
Invention Disclosure
(21) Jun Yong Kang, Hai Huang, ”Metal-free Direct Arylation of Dialkyl Phosphonates for the Synthesis of Mixed Alkyl Aryl Phosphonates”, Filed with The Board of Regents of the Nevada System of Higher Education (NSHE) on Behalf of UNLV, 62540773, August 7, 2017
(20) Jun Yong Kang, Hai Huang, ”Chiral N-heterocyclic Phosphordiamidic Acids (NHPAS) and Derivatives as Novel Brønsted Acid Catalysts”, Filed with The Board of Regents of the Nevada System of Higher Education (NSHE) on Behalf of UNLV (62521086) June 16, 2017.
(19) Huang, H.; Kang, J. Y., “Mitsunobu Reaction Using Basic Amines as Pronucleophiles” J. Org. Chem., 2017, 82, 6604-6614. DOI: 10.1021/acs.joc.7b00622
- NHP-Butane is available at Kerafast
- Most Read Articles in June July August 2017
- Highlighted in Synfacts, 2017, 13, 807. DOI: 10.1055/s-0036-1590737
(18) Moletti, N.; Kang, J. Y., “Synthesis of Diaryl-Diazaphosphonates via 1,6-Hydrophosphonylation of p-Quinone Methides with N-Heterocyclic Phosphine-Thioureas” Org. Lett.2017, 19, 958–961 DOI: 10.1021/acs.orglett.7b00261
- NHP-Thiourea is available at Kerafast
(17) Huang, H.; Kang, J. Y., “Oxidation-Reduction Condensation of Diazaphosphites for Carbon-Heteroatom Bond Formation Based on Mitsunobu Mechanism” Org. Lett.2017, 19, 544-547, DOI: 10.1021/acs.orglett.6b03709
2016
(16) Huang, H.; Palmas, J.; Kang, J. Y., “A Reagent-Controlled Phospha-Michael Addition Reaction of Nitroalkenes with Bifunctional N-Heterocyclic Phosphine (NHP)-Thioureas” J. Org. Chem.2016, 81, 11932–11939, DOI: 10.1021/acs.joc.6b02490
(15) Moletti, N.; Bjornberg, C.; Kang, J. Y. “Phospha-Michael addition reaction of maleimides employing N-heterocyclic phosphine-thiourea as a phosphonylation reagent: synthesis of 1-aryl-2,5-dioxopyrrolidine-3-yl-phosphonate derivatives” Org. Biomol. Chem.2016, 14, 10695-10704, DOI: 10.1039/C6OB01987K
(14) Moletti, N.; Kang, J. Y. “Catalyst-free synthesis of α1-oxindole-α-hydroxyphosphonates via phospha-aldol reaction of isatins employing N-heterocyclic phosphine (NHP)-thiourea” Org. Biomol. Chem.2016, 14, 8952-8956, DOI: 10.1039/C6OB01608A
(13) Huang, H.; Kang, J. Y., “Amine-Catalyzed Phospha-Michael Reaction of α,β-Unsaturated Aldehydes and Ketones with Multifunctional N-Heterocyclic Phosphine-Thioureas as Phosphonylation Reagent” Org. Lett.2016, 18, 4372–4375, DOI: 10.1021/acs.orglett.6b02121
(12) Mulla, K.; Kang, J. Y., 1,3,2-Diazaphospholidine (N-Heterocyclic Phosphine)-Mediated Carbon-Phosphorus Bond Forming One-Pot Tandem Reaction: A Route to α-amino Phosphonates, J. Org. Chem. 2016, 81, 4550-4558, DOI: 10.1021/acs.joc.6b00326
(11) Kang, J.-Y.; Johnston, R. C.; Snyder, K. M.; Cheong, P. H.-Y.; Carter, R. G. Construction of Stereogenic α,α-Disubstituted Cycloalkanones via 1° Amine Thiourea Dual Catalysis: Experimental Scope and Computational Analyses J. Org. Chem. 2016, 81, 3629-3637 DOI: 10.1021/acs.joc.6b00280
(10) Mulla, K.; Aleshire, K. L.;Forster, P. M.; Kang, J. Y. “Utility of Bi-Functional N-Heterocyclic Phosphine (NHP)-Thioureas for Metal-Free Carbon-Phosphorus Bond Construction toward Regio-, and Stereoselective Formation of Vinylphosphonates”, J. Org. Chem. 2016, 81, 77−88, DOI:10.1021/acs.joc.5b02184
- NHP-Thiourea is available at Kerafast
(9) El-Mansy, M. F., Kang, J.-Y.; Lingampally, R., Carter, R. G., Proline Sulfonamide-Catalyzed, Domino Process for Asymmetric Synthesis of Amino- and Hydroxy-Substituted Bicyclo[2.2.2]octanes. Eur. J. Org. Chem. 2016, 150–157, DOI:10.1002/ejoc.201501302![[2,2,2]-bicyclo](http://jkang.faculty.unlv.edu/wp-content/uploads/2014/06/222-bicyclo-300x75.jpg)
Invention Disclosure
(8) Jun Yong Kang, Hai Huang, Nagaraju Molleti, Functionalized Phosphonates via Michael Addition, Filed with The Board of Regents of the Nevada System of Higher Education (NSHE) on Behalf of UNLV (62376213) August 17, 2016.
2015
Invention Disclosure
(7) Jun Yong Kang, Karimulla Mulla, Methods and Compositions for Substituted Alpha-Aminophosphonates Analogues, Filed with The Board of Regents of the Nevada System of Higher Education (NSHE) on Behalf of UNLV (62309674) March 17, 2015.
2014
Invention Disclosure
(6) Jun Yong Kang, Karimulla Mulla, Kyle Aleshire, Paul M Forster, N-Heterocyclic Phosphines, Filed with The Board of Regents of the Nevada System of Higher Education (NSHE) on Behalf of UNLV (62048072) September 9, 2014.
Before UNLV
(5) Kang, J.-Y.; Carter, R. G., Primary Amine, Thiourea-Based Dual Catalysis Motif for Synthesis of Stereogenic, All-Carbon Quaternary Center-Containing Cycloalkanones. Org. Lett. 2012, 14, 3178-3181.
(4) Kang, J.-Y.; Connell, B. T., Palladium-Catalyzed Alkynylation of Secondary α-Bromo Carbonyl Compounds by the Still Coupling Reaction between sp and sp3 Carbons. J. Org. Chem. 2011, 76, 6856-6859.
(3) Kang, J.-Y.; Connell, B. T., Synthesis of Substituted Acetylenic α,β-Epoxides Followed by Indium-Catalyzed Rearrangement to 2,3,5-Trisubstituted Furans. J. Org. Chem. 2011, 76, 2379-2383.

(2) Kang, J.-Y.; Connell, B. T., Chromium-Catalyzed Homoaldol Equivalent Reaction Employing a Nucleophilic Propenyl Acetate. J. Am. Chem. Soc. 2010, 132, 7826-7827.
- Highlighted in Synfacts, 2010, 7, 1063. [link]
- Highlighted on the Organic Chemistry Portal. [link]
- Highlighted in New Methods for C-C Bond Construction, February 2011. [link]

(1) Kang, J.-Y.; Bugarin, A.; Connell, B. T., Conversion of Nitrosobenzenes to Isoxazolidines: An Efficient Cascade Process Utilizing Reactive Nitrone Intermediates. Chem. Commun. 2008, 3522-3524.
- Highlighted as a Hot Article. [link]





















